Diagnostics lie at the heart of modern healthcare, guiding nearly 70% of clinical decisions while accounting for only a fraction of total medical spending. Accurate and timely diagnostic information enables early detection of disease, informs appropriate treatment strategies, prevents unnecessary interventions, and supports effective public-health monitoring.
However, diagnostics have traditionally relied on centralized laboratory infrastructure, requiring skilled personnel, sophisticated instruments, controlled environments, and extended turnaround times. These constraints can delay clinical decision-making, limit access in remote or resource-limited settings, and create bottlenecks during outbreaks or surges in testing demand. In this context, point-of-care testing (POCT) has emerged as a transformative solution that redefines where and how diagnostic information can be generated.
Unlike conventional laboratory testing, POCT delivers results where patients are located, whether that is in clinics, emergency rooms, pharmacies, ambulances, workplaces, or even at home. This shift eliminates transport delays and reduces dependence on laboratory workflow, enabling clinicians to make real-time, actionable decisions (see Figure 1). The impact of POCT is profound: faster diagnosis of infections, earlier initiation of appropriate therapies, better chronic disease management, and greater accessibility for underserved populations.
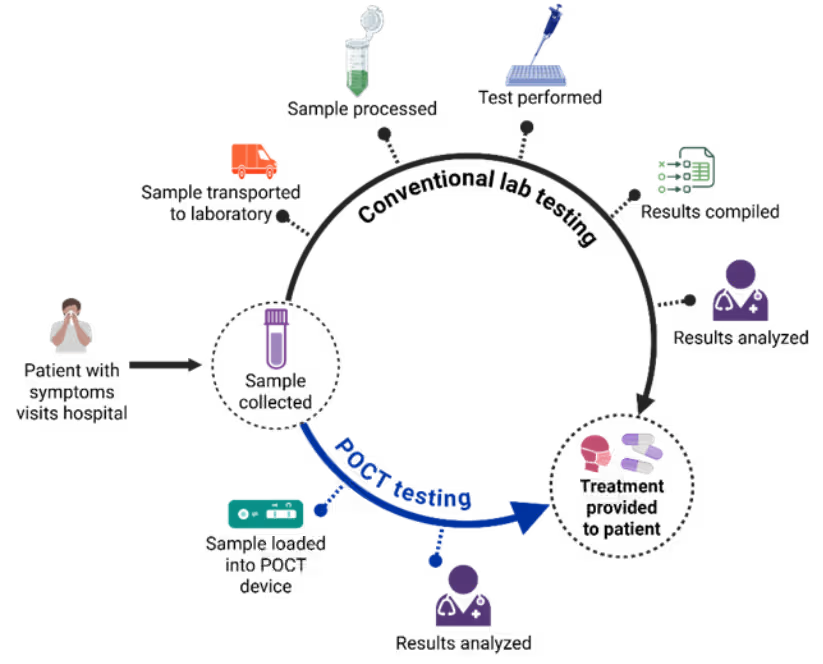
Figure 1: POCT compared to conventional laboratory testing. Illustration created with BioRender.com.
A POCT device typically integrates three functional components: sample handling, where patient specimens are collected and prepared; a detection module, which performs immunological or molecular assays using sensors such as electrochemical or optical transducers; and a readout interface, which displays results and often connects to digital platforms for real-time data sharing. These elements work together to deliver rapid and accurate diagnostics.
We are now seeing this transformation in diagnostics reflected in publication trends. We analyzed the CAS Content Collection™, the largest human-curated repository of published scientific information, using CAS SciFinder® and CAS IP Finder, powered by STNTM, and found that POCT has evolved from a peripheral topic to a major research frontier over the past decade, with a sharp acceleration during and after the COVID-19 pandemic (see Figure 2).
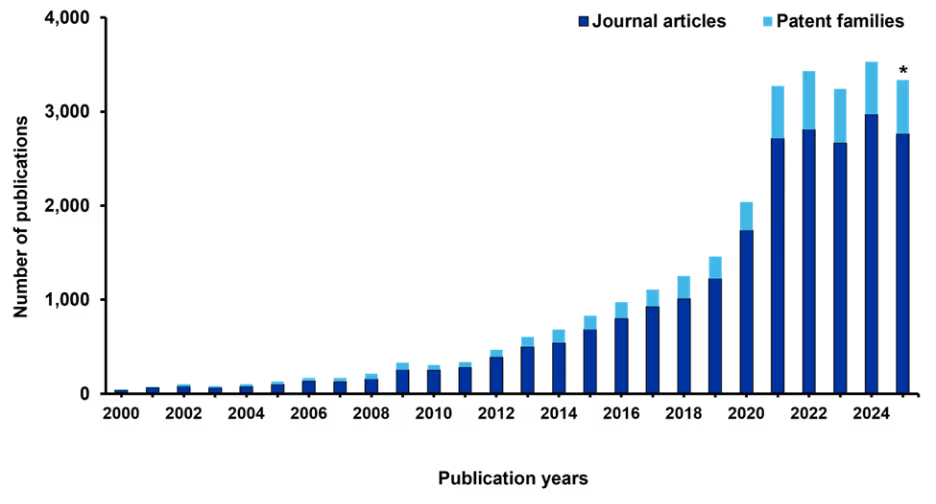
Figure 2: Publication trends in the field of POCT. Data include journal articles and patent families, defined as groups of related patents filed in multiple jurisdictions to protect the same invention. *Data for 2025 are partial through November. Source: CAS Content Collection.
Publication outputs, including journal articles and patent families, have surged dramatically, exceeding 3,000 annually in recent years. Notably, during the peak pandemic years of 2021 and 2022, approximately one-third of these publications were directly related to COVID-19, underscoring the pivotal role of POCT in pandemic response and the urgent global push for rapid, decentralized testing solutions.
Technological innovations in point-of-care testing
To ensure quality and reliability, current POCT innovations align with the updated World Health Organization REASSURED criteria: real-time connectivity, ease of specimen collection, affordable, sensitive, specific, user-friendly, rapid and robust, equipment-free, and deliverable to end-users. These attributes serve as the benchmark for modern POCT devices. Several important breakthroughs are facilitating the growth of these devices:
Platform and sensor technologies
Our publication trends analysis revealed that immunological assays dominate POCT research literature (64%), with lateral flow immunoassays (LFIA) leading significantly (44%). We saw sharp growth post-2018 driven by COVID-19 pandemic demands for scalable, self-administered rapid tests (see Figure 4A). LFIA innovations documented in the literature include enhanced sample preparation, multiplexing capabilities, and smartphone integration, reinforcing their role in decentralized testing.
Molecular POCT technologies have gained substantial traction in research publications, with PCR-based and isothermal amplification methods offering high sensitivity without complex instrumentation, while CRISPR-enabled platforms reached regulatory milestones with FDA emergency use authorizations for SHERLOCK and DETECTR in 2022, spurring hybrid LFIA-CRISPR formats.
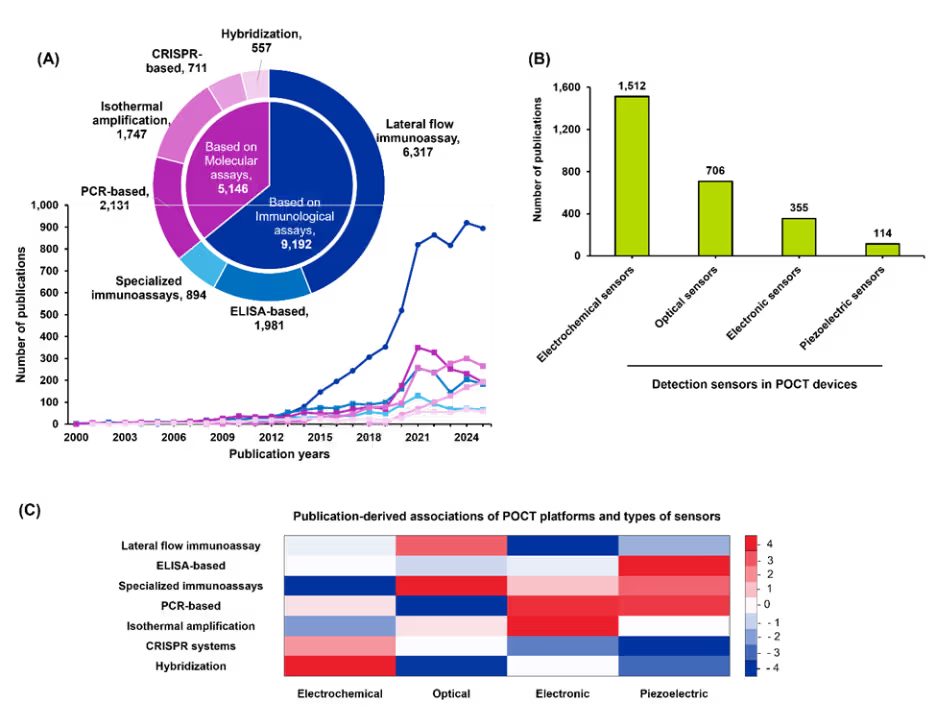
Figure 4: (A) Publication trends (line graph) and distribution of POCT platforms (pie chart) based on assay principles. (B) Relative distribution of publications on detection sensors used in POCT devices. (C) Heatmap of standardized residuals for POCT platform–disease associations. Red cells indicate over-representation and blue cells indicate under-representation relative to independence; intensity reflects the magnitude of deviation (z-score). Cells with |z| ≥ 2 were considered noteworthy deviations from independence. Data from 2000 to 2025. Data for 2025 is partial through November. Source: CAS Content Collection.
Sensor innovations mirror these assay advances, with electrochemical sensors dominating POCT device design due to their miniaturization, low power requirements, and compatibility with multiplexed detection (see Figure 4B). Optical sensors represent the second-most-researched detection method, enabling fluorescence and colorimetric detection with high sensitivity for multiplexed assays. Electronic sensors, though less prevalent in publications, provide critical digital signal processing capabilities for connected health systems, while piezoelectric sensors remain niche in research but offer unique advantages in label-free detection for ultra-sensitive applications.
To assess whether diagnostic platforms are associated with specific sensor types, we performed a chi-square test of independence based on publication frequencies, and the resulting levels of association are summarized in Figure 4C. Overall, the analysis indicates clear but moderate pairing preferences between platforms and sensors.
Electrochemical platforms tended to co-occur more often with hybridization-based sensors, and optical platforms showed a similar preference for specialized immunoassays. In contrast, electrochemical platforms were used less frequently with specialized immunoassays than expected. These patterns highlight systematic alignment and avoidance behaviors across platform-sensor combinations.
Disease-specific applications
POCT is used across disease categories, with infectious diseases leading the R&D landscape (see Figure 5A). Viral and bacterial diagnostics dominate due to their critical role in outbreak preparedness and antimicrobial stewardship, particularly during and after the COVID-19 pandemic (see Figure 5B).
Viral POCT efforts primarily focus on respiratory pathogens such as SARS-CoV-2, influenza, and RSV, but they also encompass non-respiratory conditions including HIV and hepatitis viruses. For bacterial infections, POCT platforms target sepsis-related pathogens, tuberculosis, and sexually transmitted infections like chlamydia and gonorrhea, alongside antimicrobial resistance panels for rapid susceptibility testing.
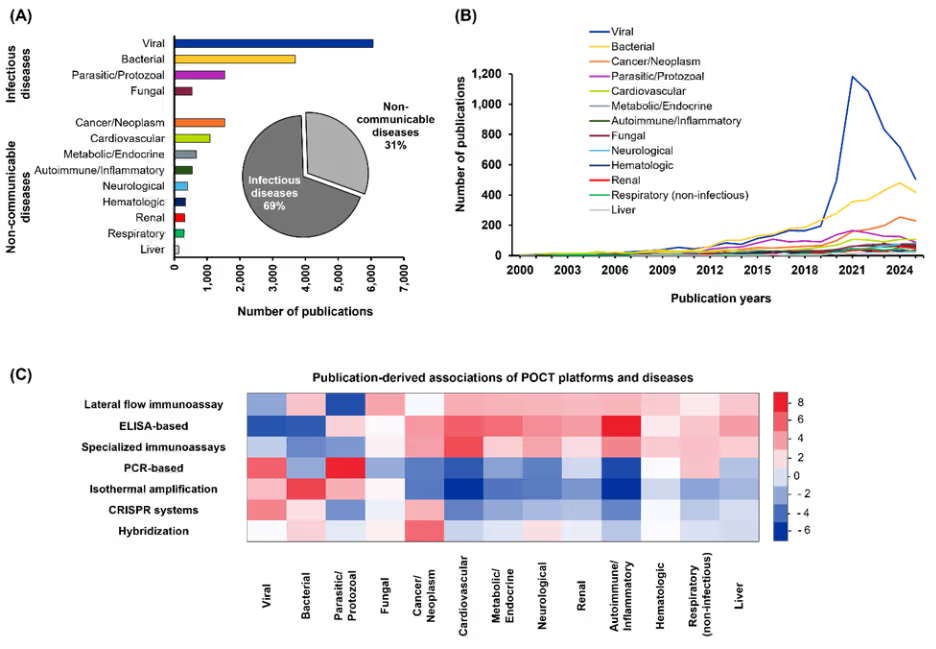
Figure 5: (A) Relative distribution of infectious and non-communicable diseases. (B) The annual publication trends for disease categories from 2000 to 2025. (C) Heatmap of standardized residuals for POCT platform–disease associations. Red cells indicate over-representation and blue cells indicate under-representation relative to independence; intensity reflects the magnitude of deviation (z-score). Cells with |z| ≥ 2 were considered noteworthy deviations from independence. Data includes journal and patent publications. Data for 2025 is partial through November. Source: CAS Content Collection.
Parasitic and fungal diagnostics remain relatively underdeveloped areas due to the lower prevalence of these conditions and limited commercial incentives, though innovations in lateral-flow and molecular assays are emerging. Cancer-related POCT ranks third and shows steady growth, driven by liquid biopsy-based approaches for circulating tumor DNA, microfluidic chips for multiplexed tumor marker detection, and electrochemical biosensors for therapy monitoring.
Beyond oncology, POCT for non-communicable diseases is expanding rapidly. Cardiovascular applications leverage high-sensitivity troponin and B-type natriuretic peptide (BNP) assays for acute care and remote monitoring, while autoimmune disease diagnostics employ portable C-reactive protein and cytokine panels for real-time inflammation profiling.
Neurological POCT is emerging through assays for Alzheimer’s biomarkers (amyloid-β, tau) and stroke-related proteins, supported by electrochemical and immunosensor platforms. Metabolic disorders such as diabetes continue to drive innovation in glucose monitoring, ketone detection, and HbA1c POCT using microfluidic and smartphone-integrated systems. Hematologic POCT includes rapid hemoglobin and coagulation assays, as well as portable platforms for sickle cell disease and anemia screening in resource-limited settings. Genetic disease POCT is advancing through portable PCR, isothermal amplification, and CRISPR-based platforms for rapid mutation detection.
To determine whether certain disease categories are preferentially linked to specific POCT platforms, we once again applied a chi-square test of independence based on publication frequencies (see Figure 5C). The results indicate clear non-random pairing patterns. Immunoassay-based POCT platforms were used far more frequently for autoimmune and inflammatory diseases, whereas PCR-based platforms were favored for parasitic and protozoal infections. Isothermal amplification methods showed a pronounced preference for bacterial diseases but were used less often for cardiovascular and autoimmune conditions.
These trends suggest distinct research preferences, with immunoassay platforms dominating chronic conditions and immune-related disorders, while nucleic acid amplification platforms are more prominent in infectious disease research.
Specimen-driven advances
Technological progress has expanded the range of biological specimens compatible with POCT. Our analysis of publication trends revealed that blood-derived samples remain the dominant choice due to their high diagnostic yield and integration with clinical workflows.
Non-invasive specimens, such as urine and saliva, have gained traction, driven by demand for patient-friendly sampling and the rise of home-based and telehealth diagnostics. These matrices enable decentralized testing without trained personnel, improving accessibility and compliance (see Figure 6A). In contrast, specimens like cerebrospinal fluid, sweat, and tears remain underrepresented because of collection challenges and limited biomarker validation (see Figure 6B).
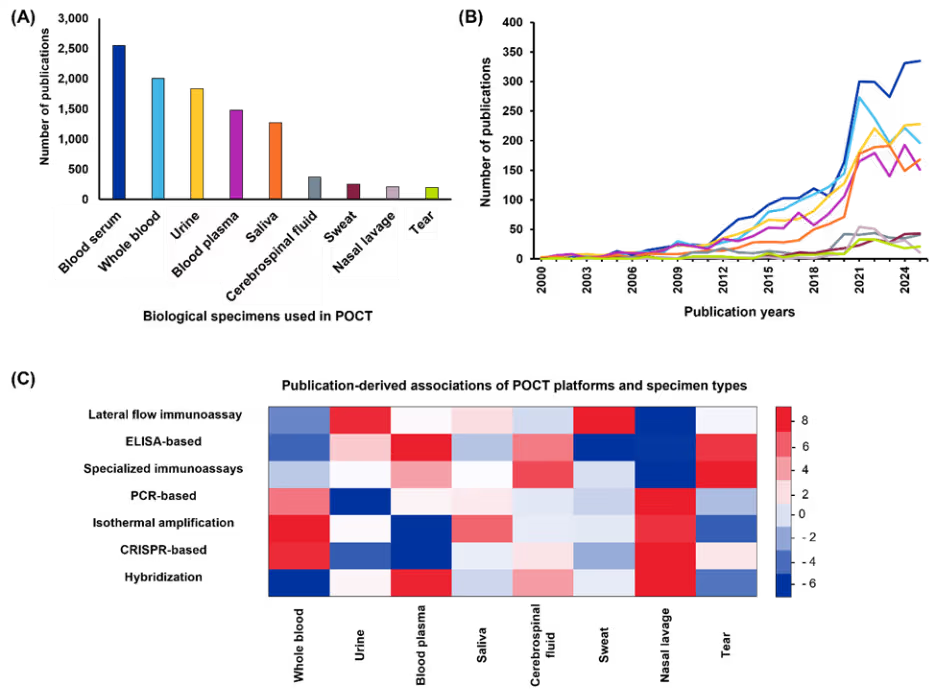
Figure 6: (A) Relative distribution of biological specimens in POCT research (B) Annual publication trends for each specimen type from 2000 to 2025 (C) Heatmap of standardized residuals for POCT platform–specimen type associations. Red cells indicate over-representation and blue cells indicate under-representation relative to independence; intensity reflects the magnitude of deviation (z-score). Cells with |z| ≥ 2 were considered noteworthy deviations from independence. Data includes journal and patent publications. Data for 2025 is partial through November. Source: CAS Content Collection.
We further examined whether publications on POCT platforms indicate preferential associations with particular specimen types (see Figure 6C). Similar to our disease-specific analysis, the chi-square test results indicated clear non-random pairing tendencies. PCR-based platforms were used far more frequently with nasal lavage and whole blood samples, while isothermal amplification methods were most common for whole blood testing.
Lateral flow immunoassays were applied across specimens but demonstrated distinct emphasis on urine and sweat, whereas ELISA-based platforms were predominantly associated with serum. These patterns reflect practical considerations in sampling feasibility, biomarker abundance, and assay compatibility reported across the literature.
Microfluidics and lab-on-a-chip integration
Microfluidics and lab-on-a-chip technologies have played a transformative role in advancing POCT platforms by enabling precise manipulation of microliter to nanoliter volumes, integrating sample preparation, reagent handling, amplification, and detection into compact devices. Our CAS publication analysis identified more than 3,000 research publications focused on microfluidics in POCT, with a steadily increasing trend over recent years.
Microfluidics has been critical for PCR-based POCT, where rapid thermal cycling, reduced contamination risk, and shortened turnaround times are essential. In addition, microfluidic elements are increasingly incorporated into advanced LFA cartridges to enable controlled fluid flow, multiplexing, and improved quantitative performance, blurring the distinction between traditional strip-based tests and chip-based diagnostics.
Centrifugal microfluidics (“lab-on-disc”) and digitally controlled droplet platforms facilitate automated analysis, including nucleic acid amplification, immunoassays, and antimicrobial resistance testing, while minimizing reagent usage. Innovations like magnetically actuated pumps enable self-contained, portable devices for low-resource environments, addressing cost and workflow integration challenges.
Digital and integrated technologies
Smartphone integration, AI, and cloud-based IoT connectivity are converging to create highly connected POCT ecosystems. Smartphone readers equipped with AI algorithms transform qualitative lateral flow results into quantitative analytics, reducing false negatives and enabling real-time data transmission to healthcare networks. Edge-AI algorithms embedded within portable POCT devices can also improve result interpretation, enable predictive analytics, and reduce bias through energy-efficient models optimized for local use. These systems support interoperability with electronic health records (EHRs) and employ secure data handling protocols to mitigate cybersecurity risks, ensuring compliance with privacy regulations.
Embedded machine learning models also enhance interpretation accuracy, automate workflows, and support predictive diagnostics, as demonstrated by UC Berkeley’s AI-driven coffee-ring assay. Collectively, these technologies deliver portable, intelligent, and connected POCT systems that combine laboratory-grade accuracy with decentralized accessibility.
Next-generation breakthroughs in point-of-care testing
POCT technologies continue to advance, particularly with the maturation of AI-driven capabilities and new types of assays:
- CRISPR-based molecular diagnostics: CRISPR/Cas systems (Cas12, Cas13) integrated with isothermal amplification and multiplexed biosensing offer ultra-sensitive detection across infectious agents, genetic disorders, and cancers.
- Smartphone-integrated biosensors: Smartphones are increasingly leveraged as integrated platforms for optical, electrochemical, and colorimetric detection, using built-in cameras or handheld potentiostat attachments.
- Pharmacogenomics and precision oncology applications: Point-of-care pharmacogenomic assays (e.g., detecting DPYD, UGT1A1, TPMT variants) guide chemotherapy dosing in real time, which can reduce drug toxicity and improve treatment efficacy.
- Wearable diagnostic devices: Devices such as adhesive patches, smart textiles, and contact lenses perform near-patient testing continuously, without requiring clinical phlebotomy, enabling rapid feedback to users and healthcare providers.
Challenges and opportunities in point-of-care testing
Despite remarkable progress, POCT faces several critical challenges that must be addressed for continued advancement:
- Regulatory harmonization: Consistency remains complex due to varying approval pathways across different regions.
- Quality assurance and standardization: POCT’s decentralized nature can compromise result consistency unless supported by robust training and quality control systems.
- Data security and privacy: These concerns have intensified with increased connectivity, requiring robust cybersecurity frameworks to protect patient information in distributed testing networks.
These challenges are formidable, but the market outlook for POCT technologies is strong. It’s projected to reach USD $78.19 billion by 2029 with a CAGR of 11.4%, largely propelled by advancements in multiplex testing platforms, portable molecular diagnostic devices, smartphone-based diagnostic tools, enhanced connectivity and data integration, and the emergence of AI-powered POCT solutions. Although individual POCT devices may incur higher per-test costs, multiple modeling studies (e.g., infant HIV NAAT, flu/RSV PCR) show that POCT often reduces overall healthcare costs through reduced hospital stays and faster treatment initiation.
As we saw in the COVID-19 pandemic, conducting testing and diagnostics outside of a traditional clinical setting delivers critical benefits to patients and public healthcare systems. By tackling the ongoing challenges to POCT development through regulatory alignment, digital security, workflow synergy, and robust quality frameworks, we can deliver faster treatments and more personalized medicine around the globe.

.avif)






