Mechanochemistry — the process where mechanical forces such as grinding, milling, or shearing drive chemical transformations — is as old as the mortar and pestle. However, the discipline is far from old-fashioned. Mechanochemical reactions are typically carried out without solvents, offering a greener approach to synthesis.
Operating under solid-state or near-solid-state conditions, mechanochemistry also enables entirely new chemical transformations. In many cases, these reactions would be inaccessible via solution-based methods. As green chemistry gains traction, this method is an increasingly important way to reduce waste and improve safety in the development of chemicals, pharmaceuticals, and more.
The surge in interest is evident in publication trends, as we found by examining the CAS Content CollectionTM, the largest human-curated repository of scientific information, accessible through CAS IP Finder, powered by STNTM (see Figure 1).
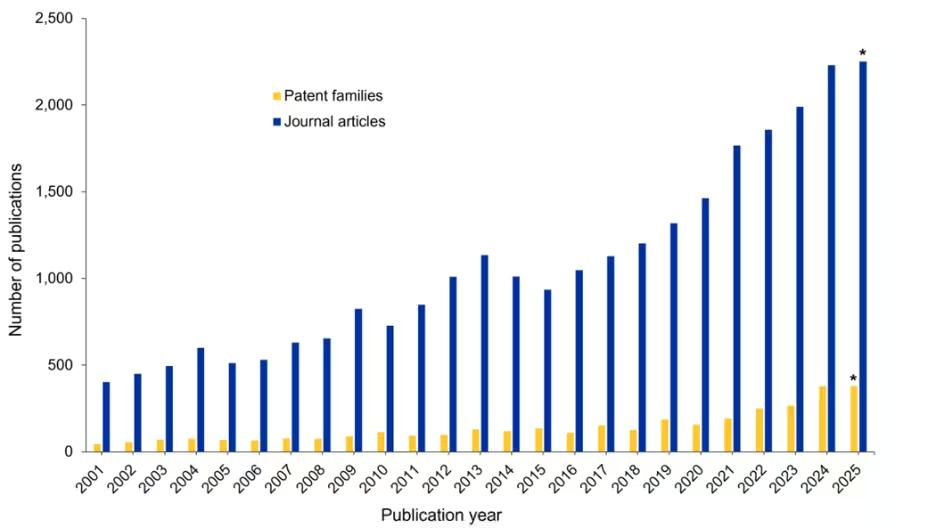
A steady increase in journal articles highlights mechanochemistry’s growing academic appeal, driven by interest in sustainable and environmentally responsible chemistry research. In contrast, the relatively low number of patent filings indicates limited commercialization, likely due to the current challenges in understanding how mechanical grinding activates different molecular groups for chemical reactions. Let’s explore how the field is evolving and its role in the larger field of green chemistry:
How mechanochemistry works
The choice of technique for a mechanochemical reaction directly impacts energy input, reaction efficiency, scalability, and sustainability, making it a critical determinant of process performance. Our analysis reveals that milling is the most prevalent technique for mechanochemical reactions (see Figure 2).
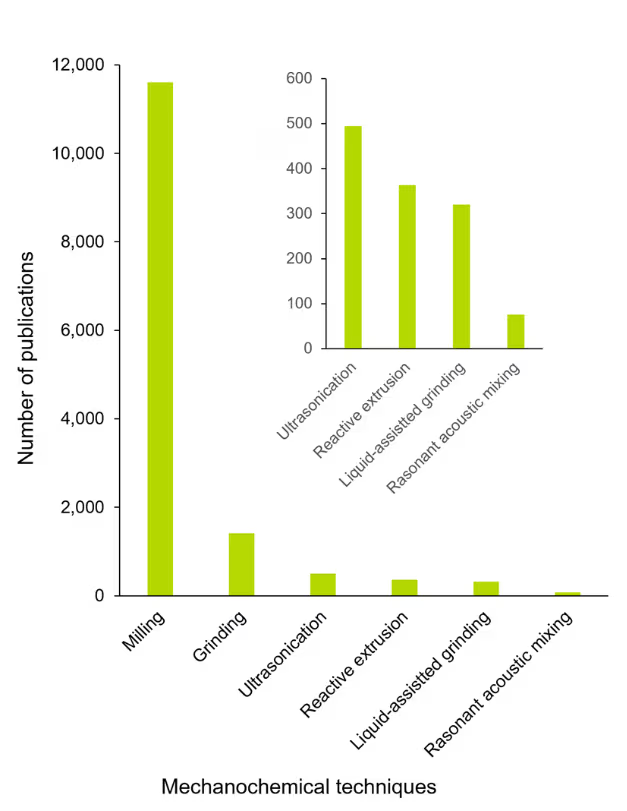
This dominance is attributed to milling’s ability to precisely control reaction parameters, such as milling frequency and the medium-to-sample weight ratio, within an enclosed system, thereby enhancing efficiency, versatility, and suitability for numerous reactions and research applications.
Mill designs are based on their motion patterns, with shaker and planetary mills being the most used. Shaker mills are typically used for small samples, where milling jars oscillate back and forth at a frequency that determines milling intensity. Planetary mills rotate jars around a central axis while spinning on their own axis, generating strong centrifugal forces that mimic gravity in industrial roller mills and provide a direct link to scale-up.
The next most common technique is grinding, which generally refers to a simpler, open-to-atmosphere mechanical process that provides limited control over reaction parameters. However, this is the cheapest and simplest approach and can be mimicked on the gram-scale with a simple mortar and pestle.
Ultrasonication uses high-frequency sound waves to drive chemical transformations through efficient agitation, enhanced dissolution, improved mass and heat transfer, and reagent sonolysis via cavitational collapse. These localized extreme conditions are effective for heterogeneous reactions, enabling them without bulk heating. This makes ultrasonication a versatile method for promoting reactions under mild overall conditions.
Newer mechanochemical techniques have appeared in a smaller number of publications:
- Reactive extrusion, particularly twin-screw extrusion (TSE), offers a scalable and continuous approach to mechanochemical processing by enabling precise control through reactor design and optimization of other continuous variables. While organic synthesis via TSE remains in its early stages, its potential is increasingly recognized. IUPAC identified reactive extrusion as one of the ten emerging technologies in chemistry with the potential to make our planet more sustainable.
- Liquid-assisted grinding (LAG) is an emerging technique that plays a crucial role in the success of mechanochemistry. By introducing a small amount of liquid (liquid additive : weight of reactants η ≈ 0-1 μL/mg), LAG enhances reactant mixing, accelerates reaction rates, improves selectivity and yield, facilitates energy transfer, and enables or directs transformations that are not achievable through neat grinding alone. Manipulating liquid additives in LAG offers a significant control on mechanochemical reactions. Additionally, LAG is efficient in screening inclusion compounds, cocrystals, polymorphs, and in organic mechanochemistry. Ion- and liquid-assisted grinding (ILAG) evolved from LAG, incorporates a small amount of salt (≤ 5 mol%) to activate the system. A related yet distinct approach is ionic liquid-assisted grinding (IL-AG), which uses an ionic liquid as the additive instead of conventional solvents or salts.
- Resonance acoustic mixing (RAM) uses low-frequency acoustic energy for mixing and inducing reactions without the need for milling media or solvents. RAM utilizes a simplified reactor design that minimizes contamination risk. It has found applications in organic synthesis and photochemical processes, offering a clean and efficient approach to mechanochemical transformations.
Mechanochemistry and advanced manufacturing across industries
Mechanochemistry has evolved into a versatile approach with applications spanning multiple domains. We analyzed concept data based on class codes and categorized them according to their respective applications (see Figure 3). Our findings indicate that mechanochemistry is widely applied in materials science, with significant contributions to energy storage, electronics, environmental applications, catalysis, and biomedical fields.
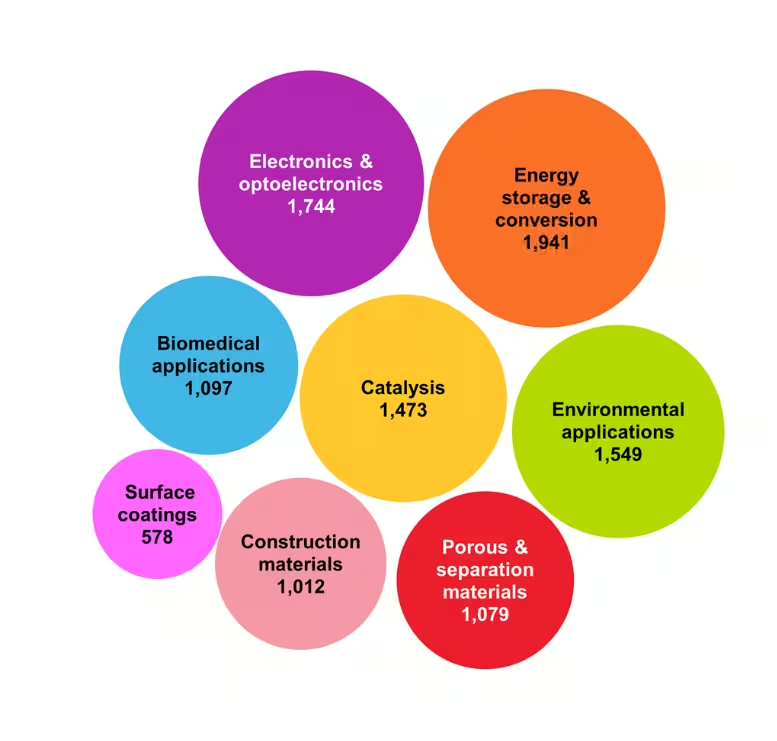
The dominance of material chemistry stems from its suitability for solid-state synthesis, eco-friendly processing, and the design of advanced materials. Additionally, historical development of mechanochemistry within mineral and materials engineering has concentrated infrastructure and expertise in these domains.
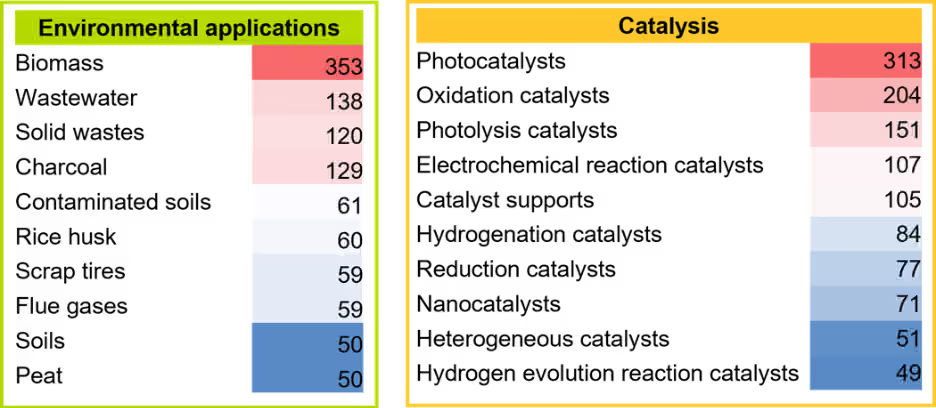
We conducted an in-depth analysis to gain comprehensive insights into each area and identified the top ten concepts within each category (see Figure 4). Solid electrolytes have emerged as the dominant focus of mechanochemistry in energy storage and conversion, as evidenced by the highest number of publications in this concept. This trend is likely driven by the growing popularity of solid-state batteries, which offer superior safety, durability, environmental friendliness, and exceptionally high energy density.
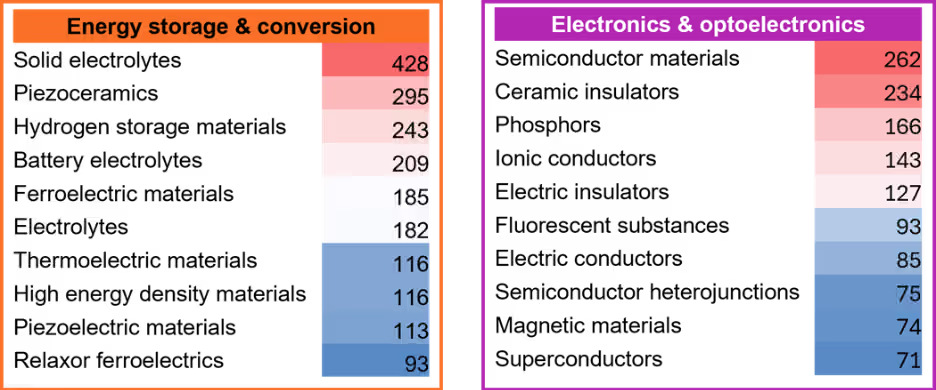


Semiconductors and ceramic insulators are dominant concepts in electronics and optoelectronics, as both are indispensable for device performance, miniaturization, and reliability. They enable high-speed, high-efficiency, and durable electronics and optoelectronics, which are central to modern technologies like smartphones, solar cells, and LEDs.
Biomass is prevalent in environmental applications because it addresses many sustainability challenges, including waste management, renewable energy production, and carbon footprint reduction. Integrating mechanochemistry with biomass valorization and processing further enhances sustainability.
In recent years, photocatalysis has emerged as a pivotal area in catalytic processes due to its sustainability and high efficiency. Building on this, mechano-photocatalytic reactions have attracted significant scientific interest as an advanced step toward greener technologies, now recognized as one of the most prominent catalytic approaches within mechanochemistry. Mechanocatalytic processes also play a crucial role in enabling diverse organic transformations.
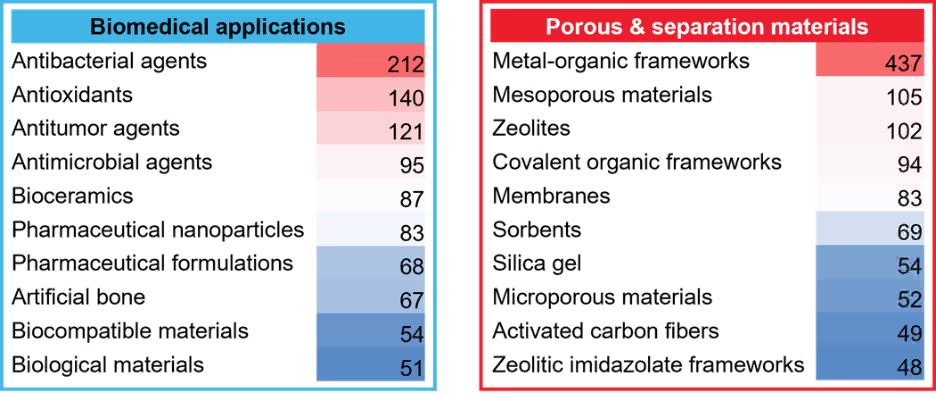
Mechanochemistry is revolutionary in biomedical applications and the synthesis of various therapeutic agents. These include pharmaceutical formulation, which involves polymorph control, co-crystal formation, and co-amorphous systems to enhance drug solubility and stability. Additionally, mechanochemistry supports nanomedicine by producing functionalized pharmaceutical nanoparticles for targeted drug delivery.
Another significant usage of mechanochemistry lies in the concept of porous and separation materials, with a predominant emphasis on metal–organic frameworks (MOFs). MOFs are distinguished by their exceptional combination of high porosity, tunable architecture, and chemical versatility, making them highly sought-after materials for diverse scientific and industrial applications. They can be synthesized in one-pot methods, making them an ideal target for mechanochemical development through the reduction of solvents.
Mechanochemistry also features applications in construction materials and surface coatings, with a predominant focus on cement and plastic films respectively. Plastic films and spray coating can also be generated from polymers produced via mechanochemical pretreatment of lignocellulosic biomass.
Making organic chemistry more sustainable
Mechanochemistry is one of the modern tools for technology-enhanced synthesis, fostering synergy between traditional organic chemistry and advanced technological innovations. Organic synthesis traditionally relies on dissolving reactants in organic solvents to facilitate reactions, which raises the question of whether such transformations can be achieved through mechanochemical approaches, thereby eliminating large volumes of solvent.
While this strategy is highly desirable from a sustainability perspective, it remains challenging because organic synthesis is deeply rooted in solution-phase techniques that ensure molecular-level precision, stereo/chemoselectivity, and stabilization. Ongoing efforts to overcome these limitations have successfully expanded the scope of mechanochemistry to include numerous organic transformations such as organocatalytic reactions and metal-catalyzed transformations, olefin metathesis, C─H activation, coupling reaction, and click reaction.
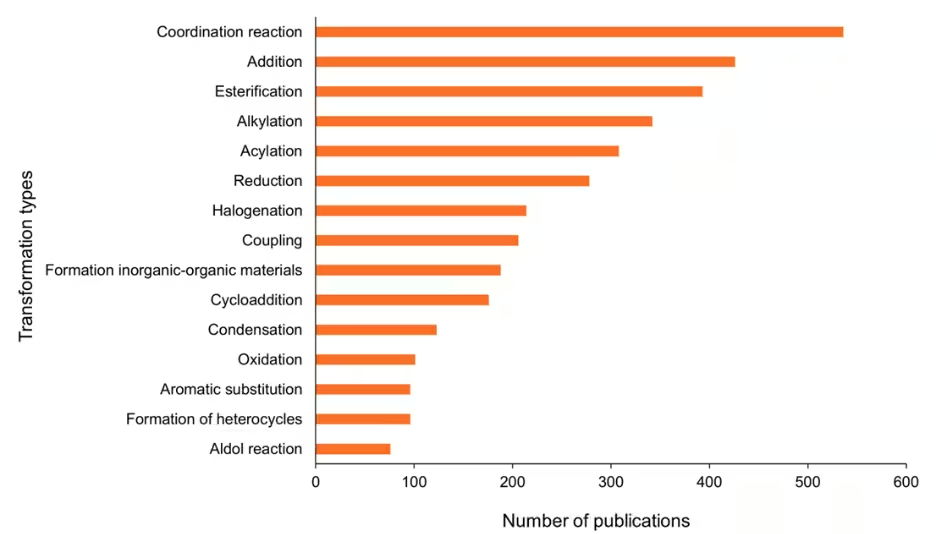
Using CAS SciFinder®, which aggregates chemistry and related science data from global sources, we analyzed reaction data and gained insights into the trends of the types of transformations now being performed with mechanochemical approaches (see Figure 5).
Researchers exploring similar trends can use CAS SciFinder to access AI-enabled search capabilities to identify patterns in organic chemistry reaction data.
Coordination reactions are the leading transformation type, closely followed by addition reactions which include the addition of alcohols/amines to aldehydes, the Michael addition, and the addition of organometallics to activated double or triple bonds. Both reaction types are inherently compatible with solid-state conditions and benefit from the intimate mixing and energy input provided by mechanical activation.
Reaction types such as esterification, acylation, and alkylation represent some of the most common functional group transformations in organic synthesis. Figure 6 illustrates the year-wise growth of the top ten reaction types, highlighting a consistent increase in the number of reactions conducted under mechanochemical conditions:
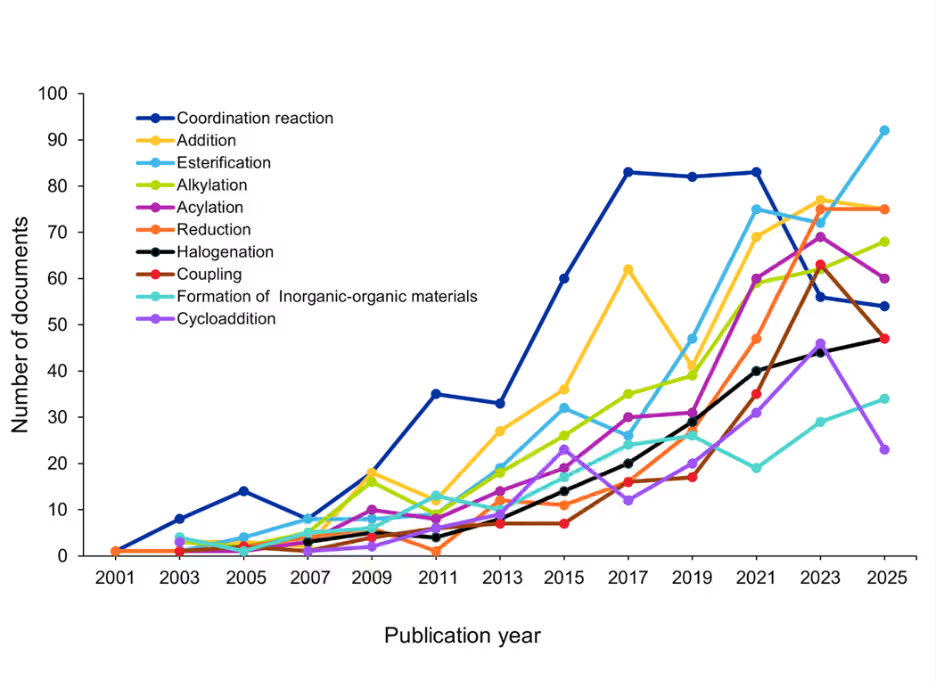
Figure 6: Growth of the top ten reaction types. Source: CAS Content Collection.
Process automation is key to scaling mechanochemistry
Mechanochemistry is increasingly recognized as a solvent-free, innovative approach to chemical synthesis. The discipline has gained widespread adoption across diverse fields, including materials science, polymer chemistry, organic and inorganic synthesis, and biochemistry. Mechanochemistry offers wide-ranging benefits, yet overcoming scalability issues is essential for translating its potential into commercial success. Consequently, efforts are underway to address these challenges through advancements in reaction methodology and mechanochemical technology development.
For example, mechanocatalysis combines mechanical force and catalysis to boost energy efficiency and achieve reactions beyond the reach of conventional solution-phase methods. It is equally impactful across domains, including organic synthesis, materials development, and polymerization. As noted, LAG is another trending approach that offers a powerful way to extend mechanochemical reactivity, facilitating reactions that are otherwise impractical under solvent-free conditions. Continuous processing is key to achieving scalability and industrialization, with extrusion-based mechanochemistry emerging as a pivotal solution. The integration of TSE technology has enabled the large-scale execution of many mechanochemical transformations.
Although mechanochemistry has achieved considerable success, a comprehensive understanding of its reaction mechanisms and theoretical foundations remains a work in progress. Therefore, focused research in this direction is essential for effective planning and successful implementation of mechanochemical processes. Moreover, integrating AI can assist in process automation and selection, paving the way for more efficient and economically viable industrial-scale mechanochemical production.
In essence, mechanochemistry is not just a trend but rather a paradigm shift toward sustainable chemical production. With continued research and technological innovation, it is poised to become a mainstream industrial process, driving progress across academia and industry.








