Executive Summary
- A May 2026 cluster of severe respiratory illnesses on cruise ships drew renewed attention to hantavirus, a rodent-borne RNA virus first identified in 1978 that causes an estimated 10,000 to 100,000 infections globally each year.
- Hantavirus produces two main syndromes: hemorrhagic fever with renal syndrome (HFRS) in Asia and Europe, with case fatality rates of 5% to 15%, and hantavirus pulmonary syndrome (HPS) in the Americas, with case fatality rates of 20% to 40%. No widely approved targeted antiviral therapy is currently available.
- Environmental change, agricultural expansion, and increased global mobility are reshaping where and how hantavirus appears, with rising case counts in South America and renewed scrutiny of the Andes virus, the only strain documented to spread person to person.
In May 2026, reports of a small cluster of severe respiratory illnesses linked to hantavirus attracted global attention, not because of their scale, but because they emerged in an unexpected setting: cruise ships. For many readers, this incident marked a first encounter with a virus that rarely appears in mainstream scientific or medical discussions. Yet hantaviruses have been recognized for decades. The virus takes its name from the Hantan River in South Korea, where it was first isolated in 1978 following earlier outbreaks of hemorrhagic fever, and it continues to cause severe disease worldwide.
Hantavirus is not a newly discovered pathogen, nor is it broadly transmissible like influenza or SARS-CoV-2. Instead, it comprises a group of rodent-borne RNA viruses that infect humans primarily through environmental exposure. Globally, an estimated 10,000 to over 100,000 infections occur annually, with the majority in Asia (particularly China and South Korea) and Europe. While the number of cases in the Americas is lower, typically hundreds per year, outcomes can be severe when infection occurs. Mortality rates vary by syndrome and geography: hemorrhagic fever with renal syndrome (HFRS), predominant in Asia and Europe, has fatality rates of 5% to 15%, while hantavirus pulmonary syndrome (HPS) in the Americas carries a disproportionately high rate of 20% to 40%. No widely approved targeted antiviral therapy is currently available.
Historically, hantaviruses have occupied a relatively specialized niche in infectious disease research, often considered regional zoonoses rather than globally relevant threats. However, evolving epidemiological patterns and unusual exposure scenarios are beginning to challenge that perception.
Understanding hantavirus: Biology, transmission, and pathogenesis
Hantaviruses belong to the Hantaviridae family and are characterized by a tri-segmented, negative-sense RNA genome enclosed within a lipid envelope (Figure 1). Each hantavirus species is typically associated with a specific rodent host, with which it has coevolved over extended periods. These reservoir hosts remain asymptomatic, allowing the virus to persist silently in natural ecosystems.
Human infection occurs primarily through inhalation of aerosolized viral particles from dried rodent urine, feces, or saliva. Following an incubation period of one to six weeks, patients initially experience non-specific symptoms including fever, muscle aches, fatigue, and dizziness. As the disease progresses, syndrome-specific manifestations emerge: severe respiratory distress in HPS or renal dysfunction and hemorrhagic complications in HFRS.
The virus exhibits limited direct cytopathic effects. Instead, disease is driven by host-mediated dysregulation of the endothelial barrier, triggering immune responses that increase vascular permeability and cause plasma leakage into surrounding tissues. In HPS, this leads to pulmonary edema and cardiovascular collapse, while in HFRS it manifests as kidney damage and hemorrhagic complications. This unique pathogenic mechanism, where disease severity reflects immune dysregulation rather than direct viral destruction, complicates therapeutic development.
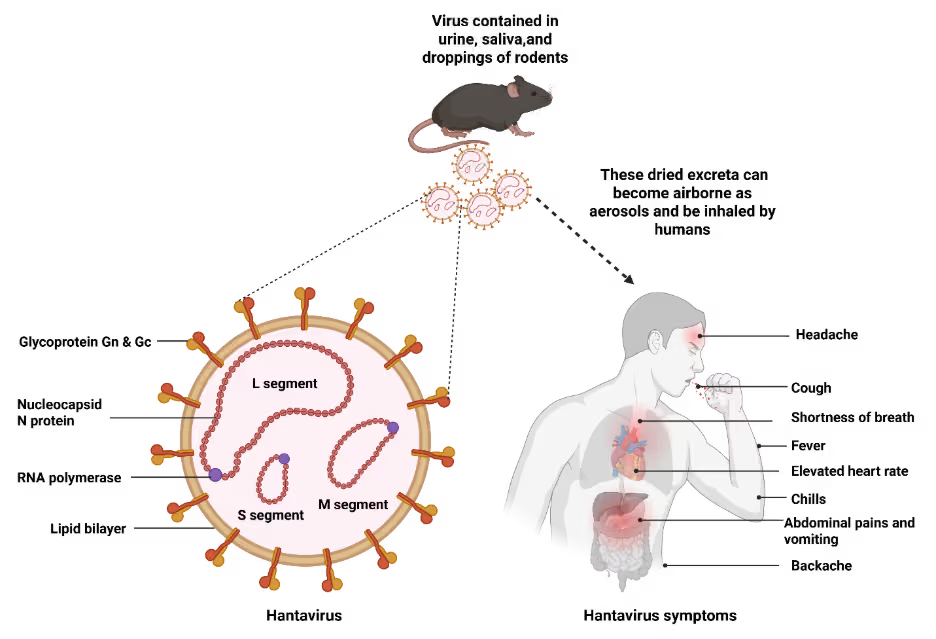
Emerging trends in hantavirus risk
Recent developments suggest a subtle but important shift in how hantavirus risk is distributed and recognized. In parts of South America, reported cases of hantavirus pulmonary syndrome have increased in recent years, with some regions experiencing higher fatality rates than historical averages. These patterns are widely linked to environmental drivers, including rainfall variability, agricultural expansion, and land‑use change, which remain the predominant determinants of hantavirus transmission through their effects on rodent reservoir abundance and human exposure.
At the same time, renewed attention has focused on the Andes virus, endemic to Argentina and Chile and notable as the only hantavirus associated with documented, albeit rare, human‑to‑human transmission. Such events typically require close and prolonged contact and represent an exception rather than a departure from the largely environmentally mediated transmission dynamics that characterize hantavirus infections globally. Recent travel‑associated investigations, including clusters linked to confined or mobile settings, have renewed attention on the potential for person‑to‑person transmission involving this strain, although inference has relied primarily on epidemiologic evidence rather than definitive virologic linkage. Efforts are currently underway to further delineate transmission pathways and assess the contribution of potential person‑to‑person spread.
Global mobility further adds to this complexity. Because the incubation period typically ranges from one to six weeks, individuals may develop symptoms far from the site of initial exposure, obscuring transmission pathways and delaying diagnosis, particularly in non‑endemic settings. Together, these factors indicate that while environmental exposure remains central to hantavirus risk, changing ecological conditions and patterns of human movement are reshaping how that risk is detected and managed.
Challenges in diagnosis and clinical management
Early detection of hantavirus infection remains difficult. Initial symptoms are non-specific, including fever, myalgia, and fatigue, and can resemble many other infectious diseases. By the time hallmark cardiopulmonary or renal manifestations develop, the disease may already be advanced. Current diagnostic approaches include detection of viral RNA via RT-PCR, serological identification of virus-specific antibodies, and immunohistochemical analysis of tissue samples in fatal cases. However, because hantavirus infections are rare, particularly in non-endemic regions, clinicians may not routinely include them in differential diagnoses which can significantly affect outcomes.
Despite sustained advances in understanding hantavirus genome organization, viral structure, and host-virus interactions, progress toward approved clinical interventions has remained limited. This is partly due to the sporadic nature of outbreaks and the complex disease mechanisms involved, in which severe manifestations are driven largely by host‑mediated vascular dysfunction rather than direct viral cytopathic effects. These factors continue to complicate diagnosis and therapeutic development.
Trends in hantavirus research: Insights from the CAS Content Collection
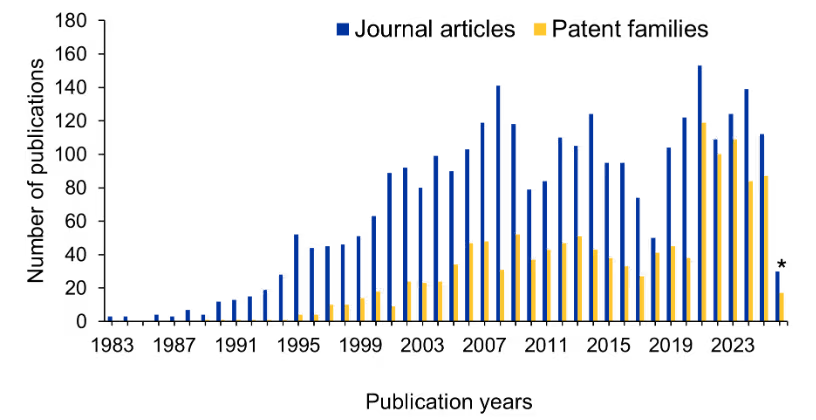
An analysis of publication and patent trends using CAS SciFinder® and CAS IP Finder, powered by STNTM tools that provide access to the CAS Content Collection™, the largest human-curated repository of published scientific information, offers an overview of research activity related to hantavirus over time (Figure 2). Overall output has remained relatively steady, with contributions from both academic and patent literature. Within the patent landscape, a notable share of filings relates to vaccines associated technologies, alongside documented activity in diagnostics, and therapeutic approaches, including antiviral agents. This illustrates continued research engagement across multiple intervention strategies for hantavirus.
Additional context is provided by Figure 3, which summarizes leading CAS indexed terms associated with journal and patent publications identified using CAS IP Finder across viral components, immune responses, and therapeutic strategies investigated in the context of hantavirus research.

Among viral components, glycoproteins, nucleocapsid proteins, and viral RNA are the most frequently studied entities, reflecting their central roles in viral entry, replication, and immune recognition. Research related to epitopes further highlights sustained efforts in antigenic characterization relevant to diagnostics, vaccine design, and immune response studies. On the host side, immune components such as immunoglobulin G (IgG), immunoglobulin M (IgM), type II interferons, and key interleukins feature prominently, reflecting continued investigation of immune responses associated with infection and disease progression.
Within therapeutic strategies, antiviral agents account for the largest share of publications, reflecting broad exploration of compounds and approaches aimed at inhibiting viral replication or modulating host-virus interactions. Research in this area has expanded beyond ribavirin to include broad‑spectrum nucleoside analogs such as favipiravir, which has demonstrated protective efficacy against multiple hantavirus species in animal models through inhibition of viral RNA‑dependent RNA polymerase activity. More recent efforts have explored host‑directed antivirals and RNA‑interference‑based approaches targeting conserved viral genome segments, although challenges related to in vivo delivery, stability, and durability remain.
Vaccine‑related research also represents a substantial proportion of the literature, reflecting ongoing evaluation of prophylactic strategies targeting viral antigens, particularly glycoproteins. Recent studies point to increasing interest in glycoprotein‑based nucleic‑acid platforms, including DNA and mRNA vaccines designed to stabilize prefusion conformations of the Gn/Gc complex, with the aim of enhancing immunogenicity and protective efficacy.
In parallel, drug delivery systems, including nanoparticle based and other advanced delivery approaches, are represented, indicating continued interest in delivery‑oriented strategies alongside antiviral and vaccine research. Publications indexed under monoclonal antibodies and neutralizing antibodies further reflect targeted therapeutic strategies focused on viral neutralization and immune mediated intervention. Fully human monoclonal antibodies isolated from hantavirus survivors, as well as polyclonal antibody preparations such as SAB 163 produced using transchromosomic bovines, have demonstrated strong post exposure protection against diverse hantavirus strains in preclinical models.
Prevention and public health strategies
In the absence of specific antiviral treatments, prevention remains the cornerstone of hantavirus control. Effective environmental strategies include rodent-proofing buildings, securing food storage, eliminating nesting sites near human dwellings, and maintaining proper waste management to reduce rodent attraction. Personal protective measures, such as using HEPA-filter respirators when cleaning potentially contaminated spaces, employing wet-cleaning techniques to minimize aerosolization, and avoiding rodent habitats, are also critical.
Public education is also essential, particularly in endemic regions. Targeted risk communication, travel advisories, and training for high-risk groups such as outdoor workers and researchers can significantly reduce exposure. In regions where the Andes virus circulates, additional precautions for close contacts of infected individuals may be warranted.
Hantavirus in a changing world
Hantavirus provides important insights into the evolving dynamics of zoonotic disease. Like other rodent-borne pathogens, it persists in natural reservoirs with periodic spillover into human populations. The emergence of strains capable of limited human-to-human transmission highlights the evolutionary potential of such viruses and underscores the importance of the One Health approach, integrating human, animal, and environmental surveillance systems.
More broadly, hantavirus illustrates the challenges posed by “neglected” emerging infections, pathogens that cause sporadic but severe disease without sustained transmission. These threats require distinct preparedness strategies focused on ecological monitoring, early detection, and strong supportive care systems.
While hantavirus is unlikely to drive large global outbreaks, its significance lies in what it reveals about shifting disease risk. Environmental change, altered rodent habitats, and increasing human mobility are reshaping exposure patterns in subtle but important ways.
Understanding hantavirus today is therefore less about tracking isolated outbreaks and more about recognizing how established viruses adapt to changing ecological and societal conditions. This is important because many cases are slow and quiet but can soon become quick and evident.
Questions and answers
What is hantavirus and how does it spread?
Hantavirus is a group of rodent-borne RNA viruses in the Hantaviridae family. People become infected primarily by inhaling aerosolized particles from dried rodent urine, feces, or saliva, often while disturbing contaminated dust in enclosed spaces. Each species has a specific rodent reservoir host that carries the virus without symptoms.
What are the symptoms and mortality rates of hantavirus infection?
After an incubation period of one to six weeks, symptoms begin with fever, muscle aches, fatigue, and dizziness. Hemorrhagic fever with renal syndrome (HFRS), found in Asia and Europe, has case fatality rates of 5% to 15%. Hantavirus pulmonary syndrome (HPS) in the Americas carries case fatality rates of 20% to 40%.
Is there a vaccine for hantavirus?
No widely approved targeted antiviral therapy or vaccine for hantavirus is currently available. Active research includes broad-spectrum nucleoside analogs such as favipiravir, host-directed antivirals, glycoprotein-based DNA and mRNA vaccines, and monoclonal and polyclonal antibody preparations including SAB-163, which has shown post-exposure protection in preclinical models.
Can hantavirus spread from person to person?
Most hantavirus infections come from environmental exposure to rodent excreta and do not spread between people. The Andes virus, endemic to Argentina and Chile, is the only hantavirus documented to transmit person to person, and only in rare cases involving close, prolonged contact.
Why are hantavirus risks changing?
Environmental drivers such as rainfall variability, agricultural expansion, and land-use change are altering rodent populations and human exposure, raising case counts in parts of South America. Global travel compounds the issue: with an incubation period of up to six weeks, people can develop symptoms far from where they were exposed.