Vector-borne diseases represent a major global health challenge, accounting for more than 17% of all infectious diseases and causing over 700,000 deaths annually. These illnesses, such as malaria, dengue, chikungunya, and Lyme disease, are transmitted by arthropod vectors, including mosquitoes, ticks, flies, and fleas (see Figure 1). Because these vectors are ectothermic (relying on external environmental sources for body temperature regulation), their life cycles are tightly linked to environmental conditions, making vector-borne disease transmission highly sensitive to climate change.
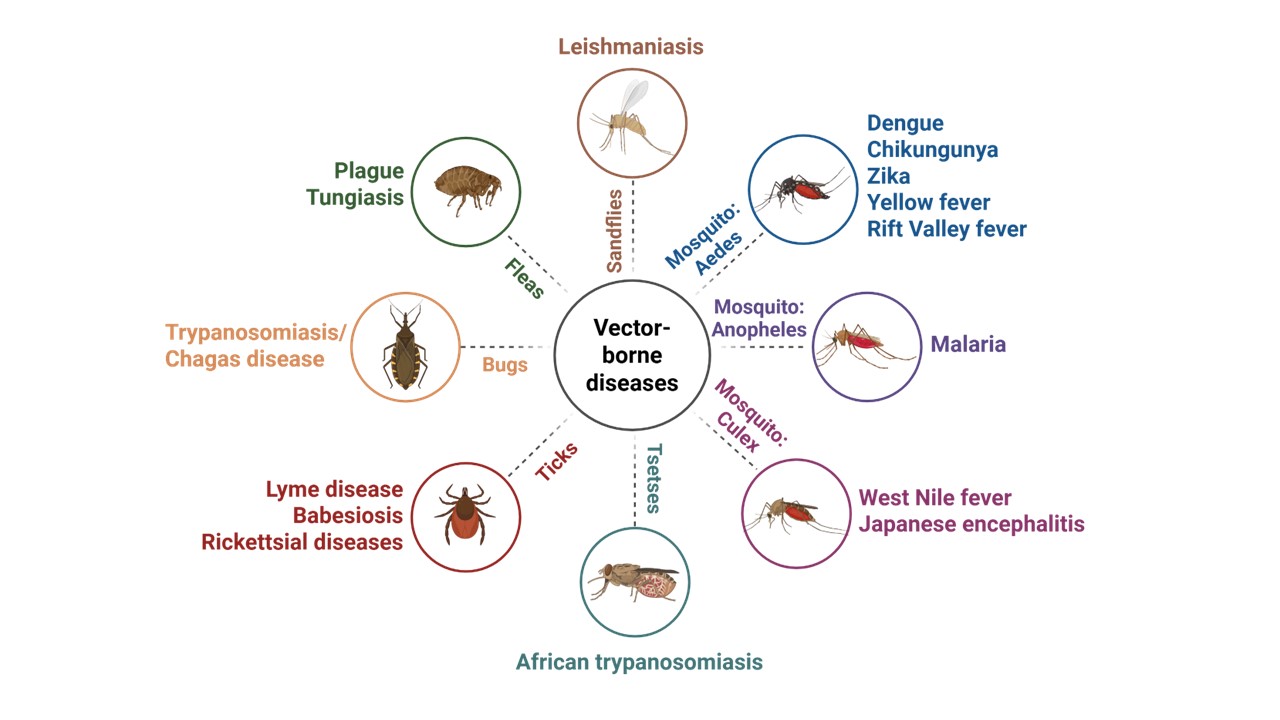
Figure 1: Overview of major vector-borne diseases and their associated vectors. Illustration created using www.BioRender.com
The Intergovernmental Panel on Climate Change (IPCC) projects global temperature increases between 1.5 and 4.5°C by 2100, with profound implications for disease dynamics worldwide. Rising global temperatures, altered precipitation patterns, and more frequent extreme weather events due to climate change are reshaping the ecological landscape that governs vector survival, reproduction, and geographic spread.
The evidence is already visible: dengue incidence has surged ten-fold in the past two decades, chikungunya has spread to over 119 countries as of 2024, and a 2021 study estimated that each year approximately 476,000 Americans are diagnosed with and treated for Lyme disease. The economic toll is equally alarming: Aedes‑borne mosquito diseases alone have an average annual cost of approximately US $1.2 billion globally.
Beyond direct healthcare costs, these diseases also impair economic development. A World Health Organziation (WHO) report notes that vector-borne diseases restrict rural and urban development in tropical and subtropical regions. They also dampen tourism — eliminating illnesses such as malaria and dengue could potentially increase global tourism spending by US $12 billion.
As climate zones shift and precipitation patterns change, it’s increasingly important to understand where disease vectors are moving and how to protect the populations at risk. The health of millions of people and economic stability for entire regions is at stake.
The research landscape of vector-borne diseases
Research interest in vector-borne diseases has surged dramatically in recent years, reflecting the growing recognition of climate-driven health risks. We leveraged the CAS Content Collection™, the largest human‑curated repository of scientific information, to map global research activity on climate change and vector‑borne diseases. We found a striking increase in scientific attention to the link between climate change and vector-borne diseases over the past two decades.
Searches were executed through CAS IP Finder powered by STNTM, which provides advanced access to the CAS Content Collection along with powerful semantic indexing and analytical tools. Using broad vector‑borne disease terminology intersected with climate‑ and environment‑related concepts, and limiting results to 2000–2025, we retrieved over 1,600 curated documents for analysis.
Our analysis revealed a >40‑fold increase in publications over the last 25 years (see Figure 2). Journal articles dominate this space, while patent activity remains minimal, with only 27 patent families (groups of related patents filed in multiple jurisdictions to protect the same invention) identified. This surge in research reflects the heightened global concern around malaria, Zika, and other vector-borne diseases, but it also underscores the difficulty in developing effective treatments, since relatively few are ready for commercialization.
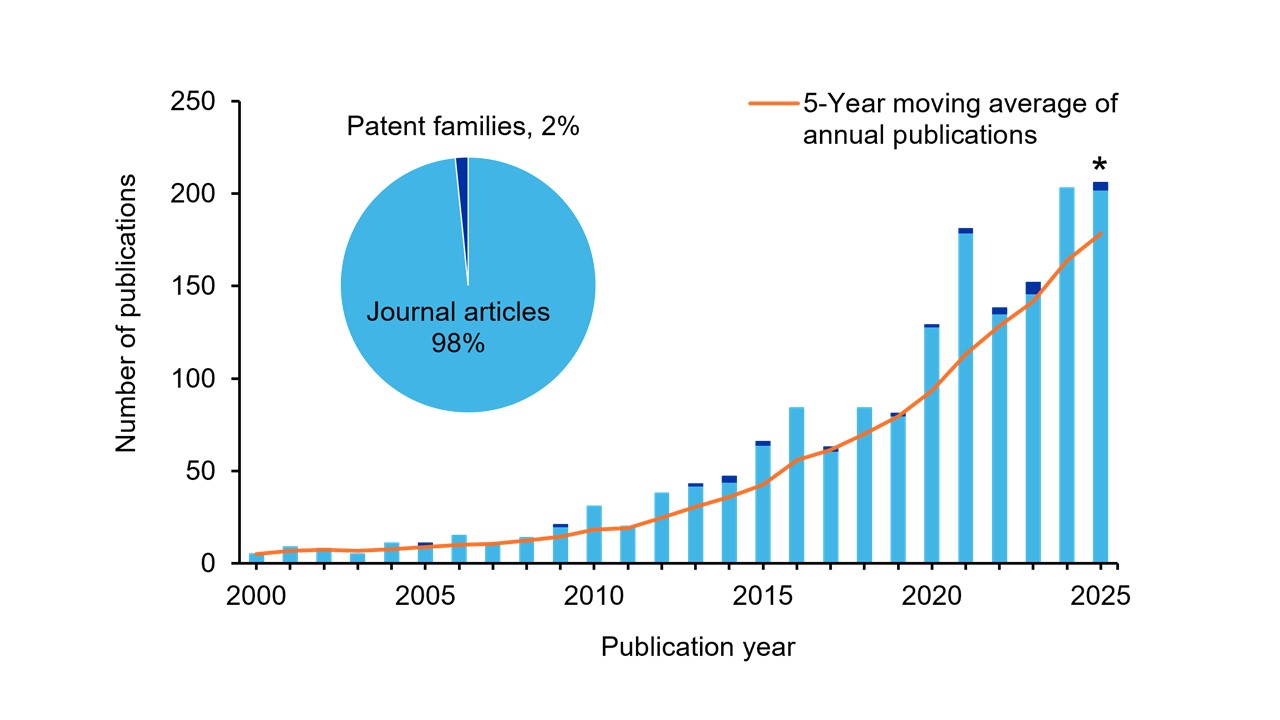
Figure 2: Annual publication trends on climate change and vector-borne diseases. Colored bars represent yearly publication counts, and the orange line shows the trailing five-year average of annual publication counts. The inset pie chart illustrates the distribution of document types. *Data for 2025 is partial through November. Source: CAS Content Collection
Using indexed concepts from the CAS Content Collection resulting from full document analysis by CAS analysts, the dataset reveals distinct patterns in the diseases, climatic factors, and vectors emphasized in the literature (see Figure 3). Our analysis shows that malaria and dengue appear to be the most-studied diseases, followed by leishmaniasis and chikungunya. Among climatic drivers, temperature emerges as the most frequently investigated factor, while humidity and precipitation also receive notable attention.
In terms of vector groups, Aedes and Anopheles mosquitoes appear to garner the most scientific interest, reflecting their role in transmitting high-burden diseases such as dengue, chikungunya, and malaria. This categorization showcases how research efforts are distributed across the climate-vector-disease interface, helping identify both well‑studied areas and gaps that warrant further investigation.
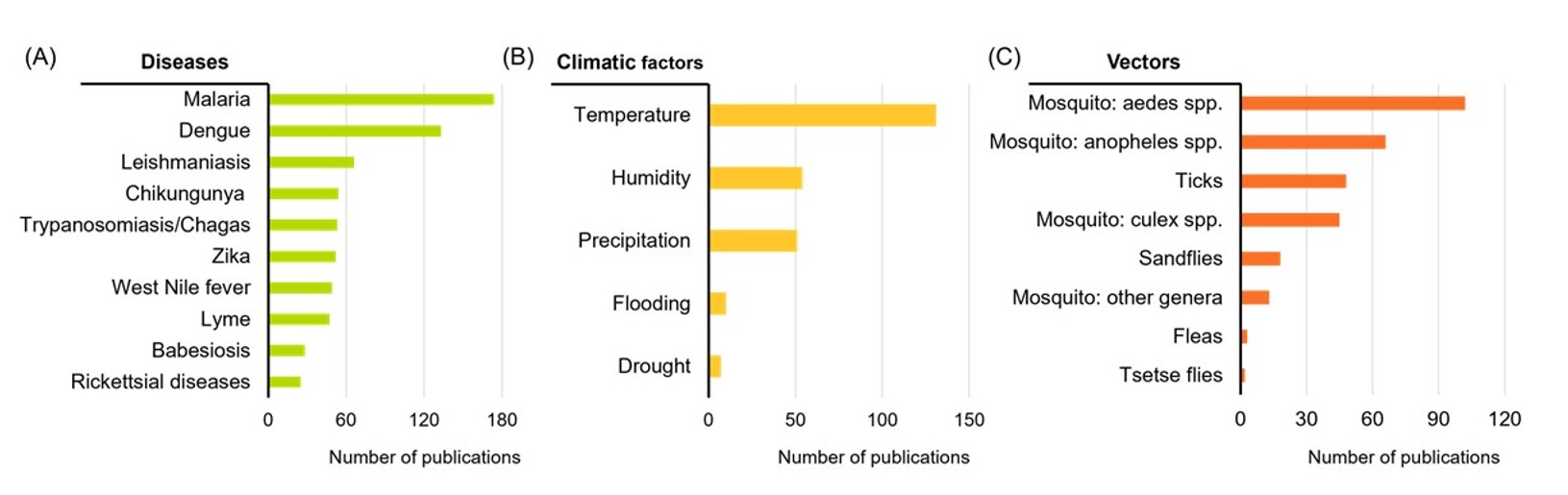
Figure 3: Relative distribution of publications on (A) major diseases, (B) climatic factors, and (C) vectors in the climate change and vector-borne diseases dataset. Data includes journal articles and patent families published between 2000 and 2025. Data for 2025 is partial through November. Source: CAS Content Collection.
A global snapshot of research activity related to climate change and vector-borne diseases is shown in Figure 4. Across the various locations, malaria, dengue, and Lyme disease appear prominently, while regional variations, such as a stronger focus on West Nile virus in the United States or leishmaniasis in Brazil and India, highlight localized research priorities. This visualization shows the global nature of vector-borne diseases and how climate-driven health conditions are impacting all areas of the world.
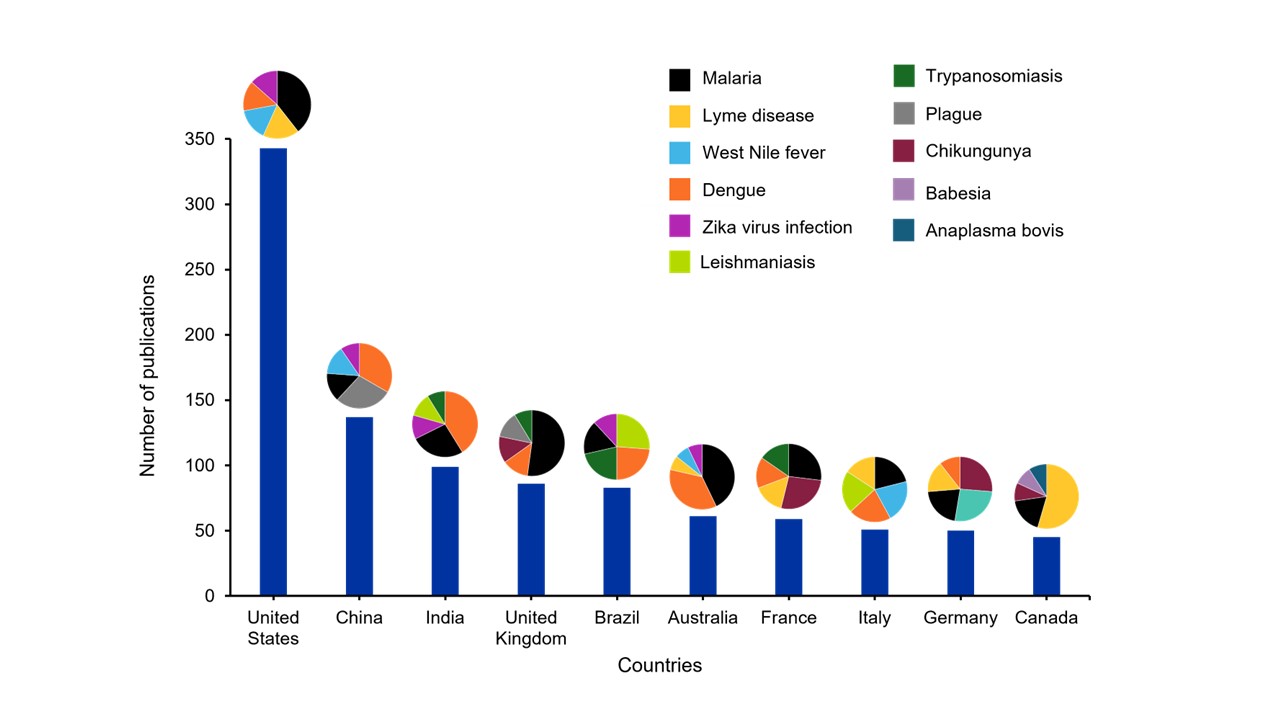
Figure 4: Publication output on climate change and vector-borne diseases across the top 10 countries. Bars indicate the number of publications, and pie charts illustrate the relative distribution across major diseases within each country’s research portfolio. Data includes journal articles and patent families published between 2000 and 2025. Data for 2025 is partial through November. Source CAS Content Collection.
Why climate change accelerates vector-borne diseases
Climate change influences vector-borne disease dynamics through interconnected environmental and biological mechanisms. These climate drivers affect ecosystems, the vectors, and human behavior, all of which create conditions conducive to disease transmission (see Figure 5).
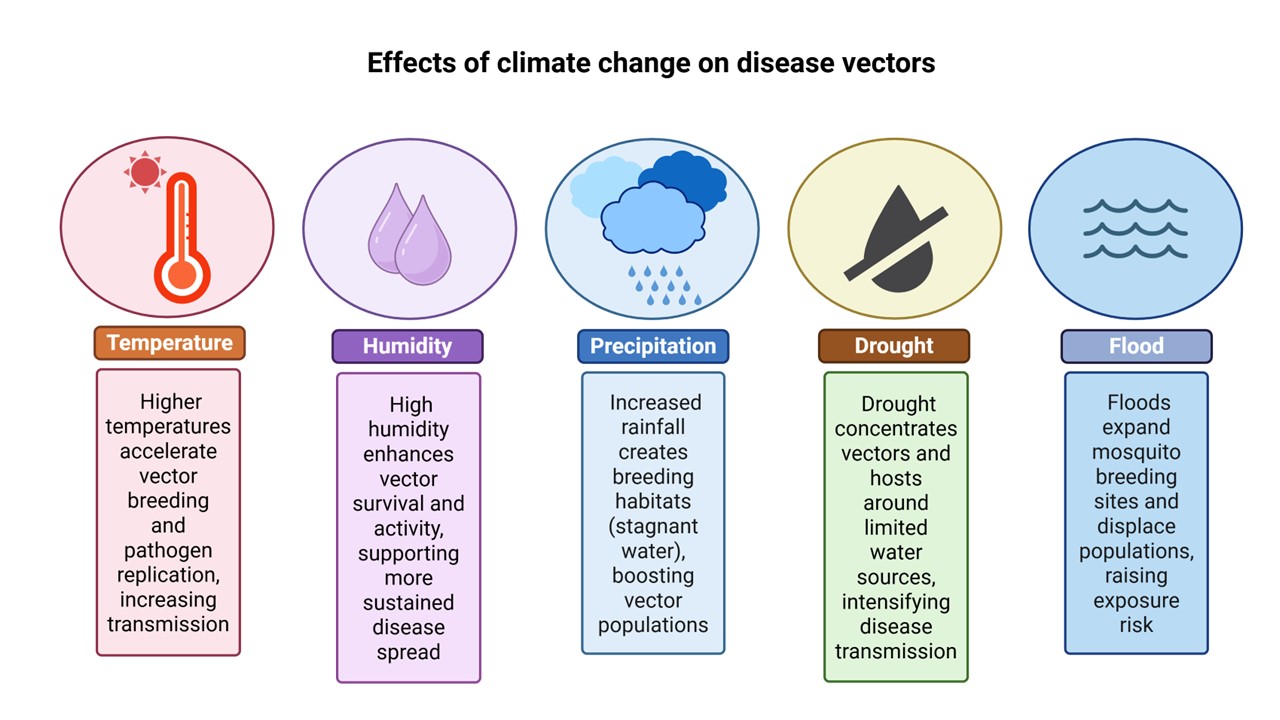
Figure 5: Climatic factors and their impact on vector-borne diseases. Created using www.BioRender.com.
Temperature effects
Temperature is the primary climatic driver of vector biology, shaping insect development, survival, reproduction, and behavior through non-linear thermal relationships. For mosquitoes, temperature influences larval development, adult longevity, blood-feeding frequency, and flight activity, with growth accelerating up to species-specific thermal optima and declining beyond.
Pathogen development within vectors, known as the extrinsic incubation period (EIP), is highly temperature-sensitive. Modeling data suggest that warmer conditions substantially shorten EIP and increase transmission potential. For example, a mechanistic model fitted to experimental datasets estimates that increasing temperature from 21 °C to 34 °C decreases the malaria parasite EIP₅₀ from 16.1 to 8.8 days. Similarly, dengue virus development in Aedes aegypti accelerates from 15 days at 25 °C to around 6.5 days at 30-32 °C (where 15 and 6.5 are means) as per temperature-dependent EIP models.
Importantly, transmission efficiency follows a “Goldilocks zone,” where moderate warming enhances risk but excessive heat becomes limiting. Dengue virus peaks at 28–32 °C, dropping sharply above 35 °C, Zika virus transmits best at 22.7°C to 34.7°C, and West Nile virus is predicted to transmit best at 23.5°C to 25.6°C. These thresholds show how climate-driven temperature shifts can expand or contract transmission windows for major vector-borne diseases, and they highlight the variability and uncertainty surrounding global warming and its exact effects on disease vectors.
Precipitation and humidity
Rainfall and humidity strongly influence vector habitats and survival, creating complex, non-linear effects on transmission. Many mosquito species depend on standing water for larval development, so moderate rainfall expands breeding sites, while extreme precipitation can flush larvae and reduce populations. Conversely, drought may eliminate habitats altogether, or concentrate vectors and hosts around limited water sources and intensify transmission, as seen during the 2012 West Nile virus outbreak in Texas, where drought amplified viral spread. Humidity further affects adult mosquito longevity and activity; low humidity reduces survival and flight, while excessive moisture can promote fungal infections, adding another layer of complexity to vector abundance.
Extreme weather events
Climate change is amplifying hurricanes, floods, and heat waves, creating unpredictable patterns in vector-borne disease transmission. Flooding can generate extensive temporary breeding habitats, fueling mosquito population surges and outbreaks of dengue, chikungunya, and Zika, such as those observed after the 2017 hurricanes Irma and Maria in the Caribbean. El Niño–driven rainfall and heat preceded major dengue surges in Peru in 2023-2024, with cases rising by over 130%. Catastrophic flooding that occurred during Pakistan’s monsoon season in 2025 triggered explosive arboviral outbreaks by disrupting water, sanitation, and hygiene systems and concentrating vector-host interactions in displaced communities. This unpredictability means that local healthcare systems worldwide must react quickly when extreme weather conditions are conducive to vector survival and disease transmission.
The spread of vector-borne diseases and potential treatments
Global dengue incidence has risen sharply over the past two decades, with record-breaking outbreaks in 2023–2024 linked to climate warming, El Niño events, and shifts in mosquito vector distribution. Mechanistic and distribution models consistently project poleward expansion and emerging seasonal suitability in temperate regions, with Europe highlighted as a major future hotspot. Temperature‑parameterized global models predict that by mid‑century, climate warming will create new regions suitable for dengue transmission, potentially exposing hundreds of millions of additional people. This expansion is driven by the ability of Aedes aegypti and Aedes albopictus to thrive in different temperature ranges, allowing both species to spread into newly warming areas. Similar climate-sensitive trends are projected for the Zika virus, with one study estimating that under high‑emission scenarios, more than 1.3 billion additional people could be at risk of Zika virus transmission by 2050, particularly in North America and Europe where populations remain vulnerable. Clinically, dengue can progress to severe dengue with plasma leakage, hemorrhage, or organ impairment, placing major strain on healthcare systems during outbreaks. Treatment remains supportive, as no widely effective antiviral therapy is currently available, and existing vaccines have limitations based on serostatus and age group. The broader Aedes‑borne arboviruses such as chikungunya and Zika also lack specific antivirals, with chikungunya causing long‑term disabling polyarthralgia and Zika associated with congenital abnormalities. Drug discovery for dengue and related arboviruses has been disappointingly slow, with few new candidates entering late-stage trials. Current strategies include broad‑spectrum antivirals, host‑targeted therapies, monoclonal antibodies, RNA-based vaccines, and emerging mRNA vaccine platforms.
Global malaria trajectories over the past decade show a mix of earlier control gains and more recent stagnation, reflected in the WHO 2024 World Malaria Report’s estimate of 263 million cases and 597,000 deaths in 2023, with stalled progress driven by vector resistance, health‑system disruptions, and emerging climate pressures. Increasingly, climate change is altering the environmental suitability for transmission, especially in transitional ecotones such as highland and fringe regions. Systematic reviews document how temperature increases and rainfall variability influence vector abundance and parasite development, with projections indicating altitudinal expansions and growing populations at risk, including in highland regions such as Papua New Guinea. A scoping review from Africa further highlights the sensitivity of Anopheles and Plasmodium development rates to even small temperature rises, while fluctuations in rainfall shape breeding‑site availability and seasonal transmission patterns. As transmission patterns shift under environmental pressures, the clinical and therapeutic challenges of malaria are becoming increasingly pronounced. Current treatments rely heavily on artemisinin-based combination therapies, yet the spread of partial artemisinin resistance poses a major threat. Preventive tools include insecticide-treated nets, seasonal chemoprevention, and the recently introduced vaccines (e.g., RTS, S, and R21), although none provide complete or long-term protection. Drug‑discovery pipelines face persistent constraints, including limited chemical diversity, increasing resistance, and the slow advancement of new therapeutic classes. In response, emerging strategies include next-generation antimalarials, liver‑stage drugs, transmission‑blocking agents, and experimental platforms such as mRNA-based malaria vaccines.
Tick‑borne diseases are showing some of the most pronounced responses to climate change, with expanding geographic ranges and rising incidence in many regions. Lyme disease, caused by Borrelia burgdorferi and transmitted by Ixodes ticks, has spread significantly across North America and Europe as warming enables tick survival in previously unsuitable areas. The complex life cycle of Ixodes, involving multiple hosts and multi‑year development, creates intricate links to climate variables. Warmer temperatures accelerate tick development and improve survival, while precipitation‑driven humidity strongly influences tick activity. Tick‑borne encephalitis (TBE) is also expanding in Europe and Asia, with new endemic foci at higher latitudes and elevations. The virus shows temperature‑dependent replication in ticks, suggesting that warming may further enhance transmission efficiency. Clinically, Lyme disease presents with a wide spectrum, from early erythema migrans (the characteristic ring-type rash) to severe neurologic and cardiac complications if untreated. However, treatment continues to rely solely on antibiotics, as no licensed human vaccine is available. For TBE, effective vaccines exist in several endemic regions, but coverage remains variable and insufficient to fully curb transmission.
These prevention and clinical gaps reflect broader innovation challenges in tick‑borne disease management, where therapeutic pipelines remain sparse, and advances in biomarker‑guided diagnostics or curative interventions remain limited. Consequently, research is shifting toward new approaches beyond traditional pathogen‑directed antibiotics. Promising strategies under exploration include anti‑tick vaccines designed to disrupt vector feeding, biological vector‑control approaches targeting tick populations, host‑directed therapies to mitigate disease severity, and emerging vaccine platforms such as mRNA technologies. Additionally, antisense oligonucleotides (ASOs) are being investigated as precision molecular tools for suppressing pathogen gene expression, though these approaches remain in early development.
Economic dimensions: From burden to investment opportunity
Vector-borne diseases impose significant direct costs on healthcare systems, and climate change amplifies these pressures by driving outbreaks into regions with limited preparedness. For example, dengue treatment costs per episode range from US $15 for outpatients to over US $9,300 for intensive care unit patients. As dengue spreads into temperate, high-income regions, the per-case economic burden rises sharply due to higher healthcare costs and longer hospital stays. Conversely, in developing regions, vector-borne illnesses consume a significant portion of public health budgets that could otherwise be directed to different essential health services.
The impact extends beyond healthcare systems. Tourism-dependent economies are highly vulnerable to lost revenue due to higher disease incidence. As seen during the 2015–2016 Zika epidemic, Latin America and the Caribbean lost an estimated US $7-18 billion in tourism revenue per a United Nations Development Programme report.
Studies in African farming communities report substantial productivity losses; households may lose nearly 28% of their seasonal income due to malaria, and crop yields are consistently lower when malaria incidence is high.
This data highlights the importance of prevention when confronting vector-borne diseases. Economic analyses consistently show that preventive systems yield far greater returns than reactive outbreak response. Climate‑informed early‑warning systems for vector‑borne diseases have substantially enhanced the timeliness and effectiveness of public‑health interventions, reducing the scale, severity, and associated costs of outbreaks by enabling earlier action. Likewise, economic evaluation frameworks indicate that responses triggered by early‑warning intelligence provide positive net benefits for health systems, reiterating their value as cost‑effective tools for outbreak preparedness.
Investment in preventive systems could therefore drive extensive economic growth in the regions most affected by vector-borne diseases today, and they could avoid economic shocks in areas where these insects and the diseases they carry will move to as climate change continues.
Future directions: Adaptive strategies and research priorities
Climate-driven vector-borne disease risks demand integrated, forward-looking strategies that combine surveillance, prevention, and resilient health systems. Preparing for multiple climate scenarios is critical, as moderate warming (+2 °C) may expand vectors into temperate zones, while extreme warming (+4 °C) could reshape disease dynamics entirely.
Key priorities for public health systems include AI-powered early warning systems integrating climate and health data; climate-adapted vector control through integrated vector management; novel tools like gene-drive mosquitoes; accelerated development and deployment of vaccines, therapeutics, and diagnostics; and building climate-resilient health systems with strong supply chains and digital decision support. Digital decision support includes dashboards, predictive analytics platforms, and automated alerts that help health authorities interpret surveillance and climate information in real time and guide timely, evidence-based public‑health responses.
Policy frameworks must embed climate-driven health planning in national adaptation strategies and strengthen global cooperation. Research should focus on vector-pathogen-climate interactions, predictive modeling under diverse scenarios, evaluation of genetic and biocontrol technologies with robust safety and ethics, and social dimensions such as community engagement and equitable implementation.
The unpredictability of climate impacts on insects, their life cycles, and the pathogens they carry make these developments challenging, but the severity of vector-borne diseases and their economic impacts demand action.








