Gain new perspectives for faster progress directly to your inbox.
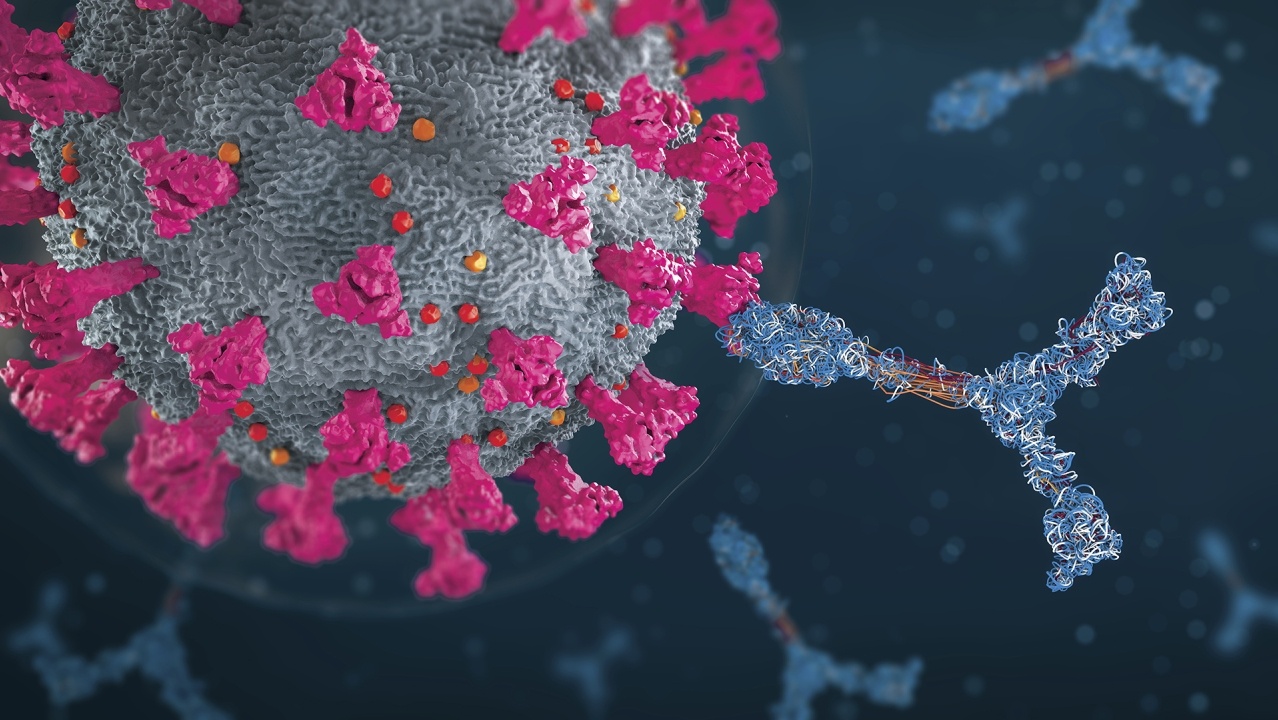
The last year has seen an unprecedented number of vaccine candidates directed at the COVID-19 pandemic. As of the end of February 2021, several vaccines had been conditionally approved, and others are close to such approval. It is likely that many more still in clinical trials will come to market in the next few years.
This report examines these vaccines and the related research effort, both traditional and forward-looking, to illustrate the advantages and disadvantages of their technologies, to denote the use of adjuvants and delivery systems in their application, and to provide a perspective on their future direction.